Read on to learn more about the Pfizer COVID-19 vaccination, and if you still have questions you can visit the Department of Health website at or consult your local community pharmacist or other trusted healthcare provider. Delaying the second dose by up to twelve weeks increases. Researchers found that the vaccine was 95 percent effective at. The World Health Organization (WHO) recommends an interval of three to four weeks between doses. The Pfizer vaccine has gone through rigorous evaluation and approval processes, and both the Therapeutic Goods Administration (TGA) and the Australian Technical Advisory Group for Immunisation (ATAGI) have approved its use for all those aged 12 years and over, as well as those who are pregnant. The Pfizer-BioNTech clinical trial evaluated vaccine effectiveness 1 week after participants had gotten their second dose. Centers for Disease Control and Prevention, the second dose. 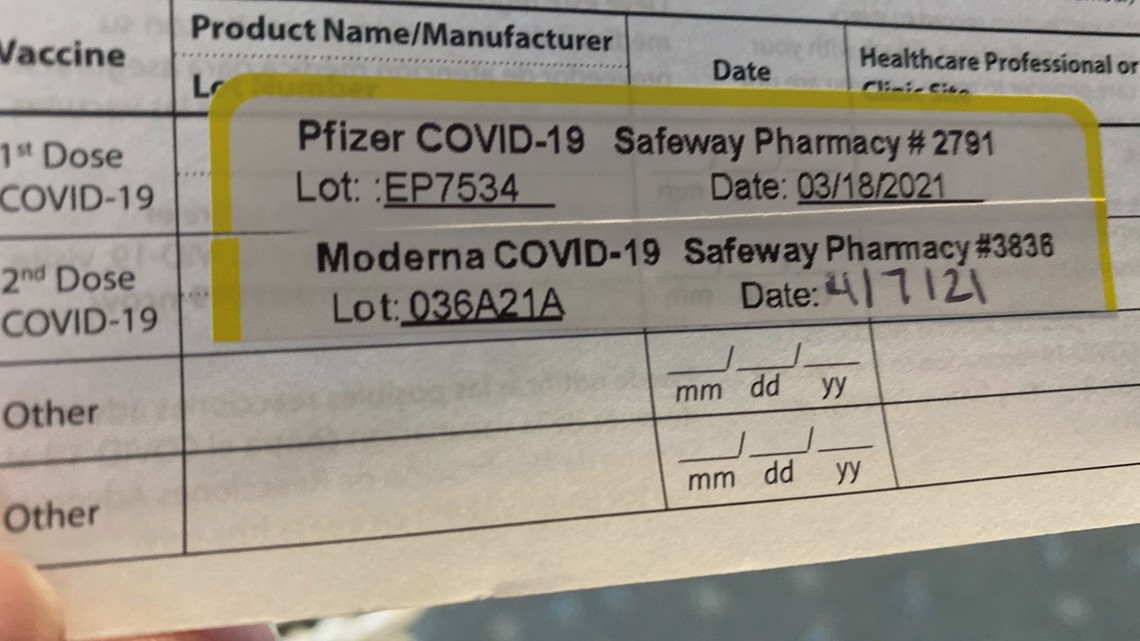
The Centers for Disease Control and Prevention quietly changed its. Covid-19 vaccines in Tucson, Arizona, U.S., on Friday, Jan. 
We’ve put together some helpful FAQs to help, but in short, the Pfizer vaccine is very similar to the Moderna vaccine, in that both are mRNA vaccines that work by helping your immune system create new antibodies that help fight off the virus that causes COVID-19. In its clinical trials, Pfizer administered the second dose 21 days after the first. Bins of syringes for the Pfizer BioNtech and Moderna Inc. A total projected 9 million doses of the Pfizer COVID-19 vaccine arrived on Australian shores in October, and although the Pfizer vaccine is well known on the market, we’re sure you have a few questions, and it’s great to be informed.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
